
Smedley, in The Interpretation of Ionic Conductivity in Liquids (Plenum Press, London/New York, 1980).ĭ. Spiro, in Physical Chemistry of Organic Solvent Systems, Chap. Stokes, in Electrolyte Solutions, 2nd edn. Francks, in Water (Royal Society of Chemistry Paperbacks, London, 1984). Glycerol is a trihydric alcohol, of 15.0 Poise viscosity at 20 ☌, miscible with water and ethanol in all proportions, and is immiscible with hydrocarbons. 
Ives, Consultant Bureau, New York, 1965).į. 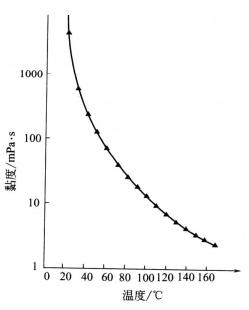
Samoilov, in Structure of Aqueous Electrolyte Solutions and the Hydration of Ions ( Translated by D. Dalton, in Glycerol (American Chemical Society Monograph Series, 1953). Using previously measured transference numbers for KCl and NaCl in glycerol, values of limiting Walden products for the individual alkali-metal and halide ions in glycerol have been derived and compared with those in aqueous and other alcohol solutions. Values of the molar conductivity at infinite dilution were obtained by extrapolation using the conductance equation of Onsager. The electrical conductivities of solutions of salts (KCl, NaCl, KBr, NaBr, NaI, KI, and LiBr) in glycerol have been measured at three concentrations (approximately 0.01, 0.1, and 0.3 M) at 25☌. Specific agreement between theory and experiment was not generally good. Viscosity B-coefficients are compared with those calculated applying existing theories based on the model of hard-charged spheres moving in a solvent continuum. The Kaminsky method, based on reference electrolyte (on B K+ = B Cl-), was used in glycerol. Heating of glycerin from room temperature to 100C decreases its viscosity by two orders of magnitude, thus allowing one to increase the Reynolds number by the same factor, the density and. The viscosity data were analyzed by means of the Jones–Dole equation. The Debye–Hückel limiting law was assumed to be valid at low concentrations, and values of the molar volumes at infinite dilution were obtained by extrapolation. 5 IT5/1.5 SDS in water glycerin mixed solvents at. The apparent and partial molar volumes ( V Φ and V 1) in mixtures of KCl, NaCl, KBr, KI, and glycerol were calculated from the density data. (a) and storage modulus G (b) versus frequency for samples of. This paper presents the results of density, viscosity, and electrical conductivity measurements for glycerol solutions of some alkali-metal halides at 25☌.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
